Chain of Custody in Medical Specimen Transport: A Complete Guide

In medical specimen transport, chain of custody is the documented, unbroken record of every person and process that handles a specimen from the moment it is collected to the moment it is accessioned at the laboratory. It serves two essential functions: it ensures regulatory compliance under HIPAA, CLIA, and CAP requirements, and it provides the traceability framework needed to prevent errors, resolve disputes, and protect diagnostic integrity. A gap in the chain of custody is not an administrative inconvenience. It is a clinical and legal liability.
For healthcare organizations managing high-volume outreach specimen logistics, chain of custody documentation must be more than a signature on a paper log. It must be a digital, real-time system that captures every handoff, records environmental conditions, tracks location, and generates auditable records that satisfy the most rigorous regulatory inspections. The difference between a chain-of-custody-focused courier and a general delivery service is the difference between verified traceability and assumed accountability.

1. What Chain of Custody Means in Healthcare Logistics
Chain of custody in healthcare logistics encompasses far more than proving a specimen was delivered. It documents who collected the specimen, who packaged it, who picked it up, what conditions it was transported under, who received it at the laboratory, and what its condition was at every transition point. Each handoff represents a potential failure point where mislabeling, misrouting, temperature compromise, or loss of accountability can occur.
In legal and forensic contexts, chain of custody has always been a strict documentation requirement. In clinical diagnostics, the stakes are equally high. A contested diagnostic result, whether in oncology, toxicology, or infectious disease, may require documented proof that the specimen was handled properly throughout its entire journey. Without that documentation, the result itself becomes indefensible, regardless of the analytical quality of the laboratory.
For laboratories managing outreach programs, chain of custody is also a competitive differentiator. Referring providers increasingly evaluate their laboratory partners not just on turnaround time and test menu, but on the quality and transparency of the logistics operation that connects them. A laboratory that can provide real-time tracking and documented chain of custody builds client confidence that one relying on paper-based tracking cannot match.
Chain of Custody Encompasses:
- Specimen identification and condition verification at the point of collection
- Documented handoff from collection site to courier with timestamps and electronic signatures
- Environmental condition monitoring throughout the transport duration
- GPS-tracked courier location and route documentation
- Delivery confirmation with condition assessment at the receiving facility
2. Regulatory Requirements for Specimen Traceability
Multiple regulatory frameworks require chain of custody documentation for medical specimen transport, each with specific requirements that logistics operations must satisfy. HIPAA mandates that protected health information associated with specimens remains secure and traceable throughout transport. CLIA and CAP accreditation standards require laboratories to demonstrate control over the pre-analytical phase, including transport conditions and documentation. State health departments may impose additional requirements specific to their jurisdictions.
During accreditation inspections, auditors routinely request documentation demonstrating that the laboratory maintains control and visibility over specimens from collection through accessioning. A CLIA and CAP compliant transport operation must produce records showing who handled each specimen, when each handoff occurred, what environmental conditions were maintained, and whether any exceptions or deviations were identified and addressed.
For HIPAA-compliant medical courier operations, chain of custody documentation must also demonstrate that patient information was protected at every transition point. This includes secure handling of requisition forms, barcoded specimen identification that prevents unauthorized access to patient data, and secure digital systems that restrict access to authorized personnel only.
Regulatory Documentation Requirements:
- HIPAA: Secure handling and traceability of all materials containing protected health information
- CLIA: Documentation of pre-analytical specimen conditions and handling procedures
- CAP: Audit-ready records demonstrating laboratory control over the entire specimen lifecycle
- State regulations: Jurisdiction-specific transport documentation, licensing, and reporting requirements
- DOT: Hazardous materials documentation for specimens classified as Category B or requiring dry ice transport
3. Digital vs Paper-Based Chain of Custody
Paper-based chain of custody systems persist in many healthcare logistics operations, but they introduce significant risk. Paper logs can be lost, damaged, illegibly signed, or incompletely filled out. They provide no real-time visibility, cannot trigger automated alerts when exceptions occur, and require manual retrieval and review during audits. Every paper-based handoff is a potential gap in the documented chain.
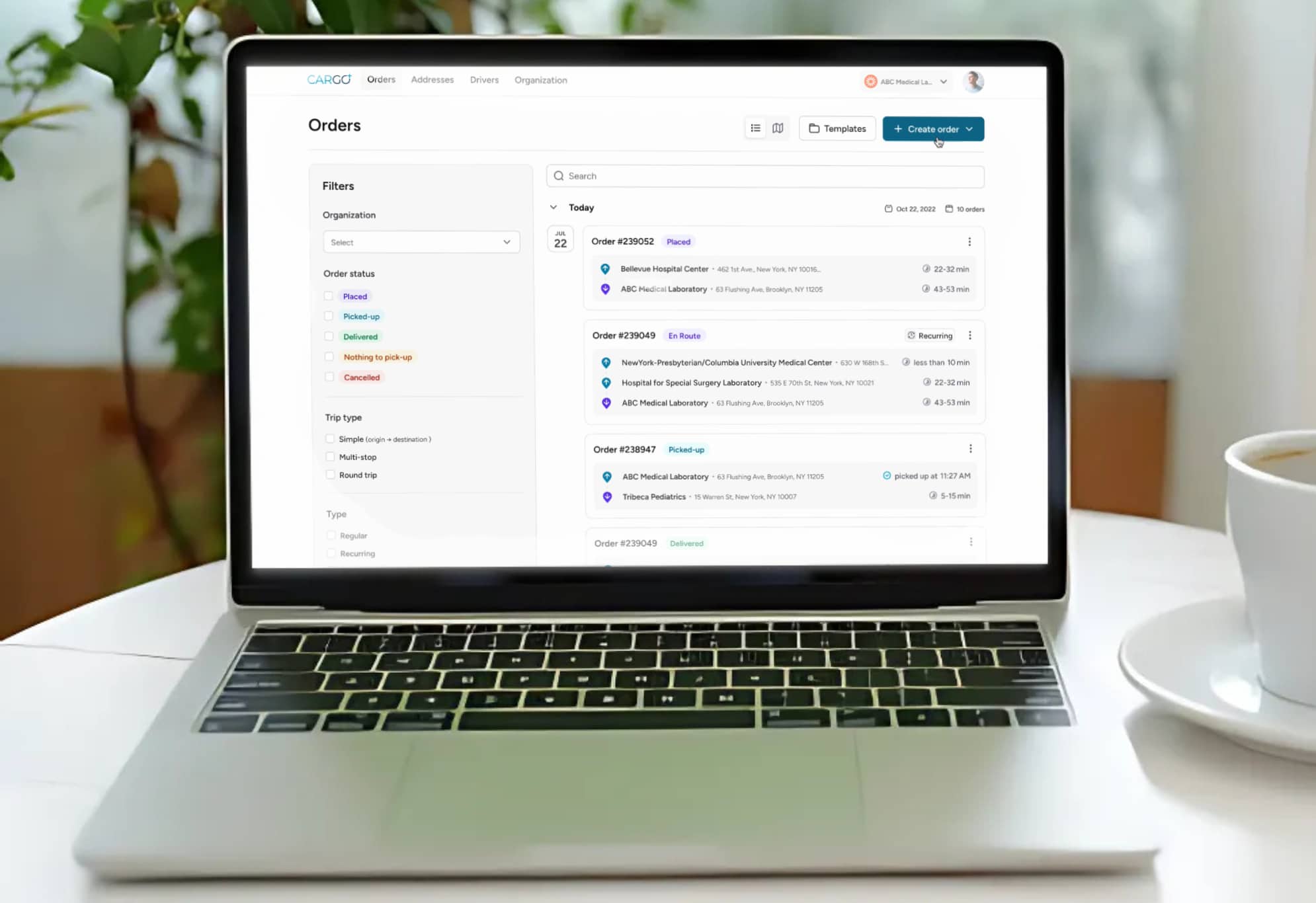
Digital chain of custody systems eliminate these vulnerabilities. Electronic signatures with timestamps provide unambiguous documentation of every handoff. Barcode or QR-based specimen identification prevents manual entry errors that can lead to mislabeling or misrouting. Real-time tracking provides continuous location visibility, not just pickup and delivery confirmation. And automated audit trails generate the documentation needed for regulatory inspections without manual compilation.
The transition from paper to digital chain of custody is not merely a technology upgrade. It is a fundamental improvement in traceability quality that reduces error rates, strengthens compliance posture, and provides healthcare providers with operational visibility that paper-based systems cannot deliver.
Digital Chain of Custody Advantages:
- Electronic signatures with precise timestamps eliminate ambiguity in handoff documentation
- Barcode and QR scanning prevent specimen misidentification and manual entry errors
- Continuous GPS tracking provides real-time location data, not just pickup and delivery confirmation
- Automated exception alerts notify stakeholders of route deviations, temperature excursions, or missed time windows
- Instant audit trail generation eliminates manual record compilation for regulatory inspections
4. Technology Infrastructure for Chain of Custody
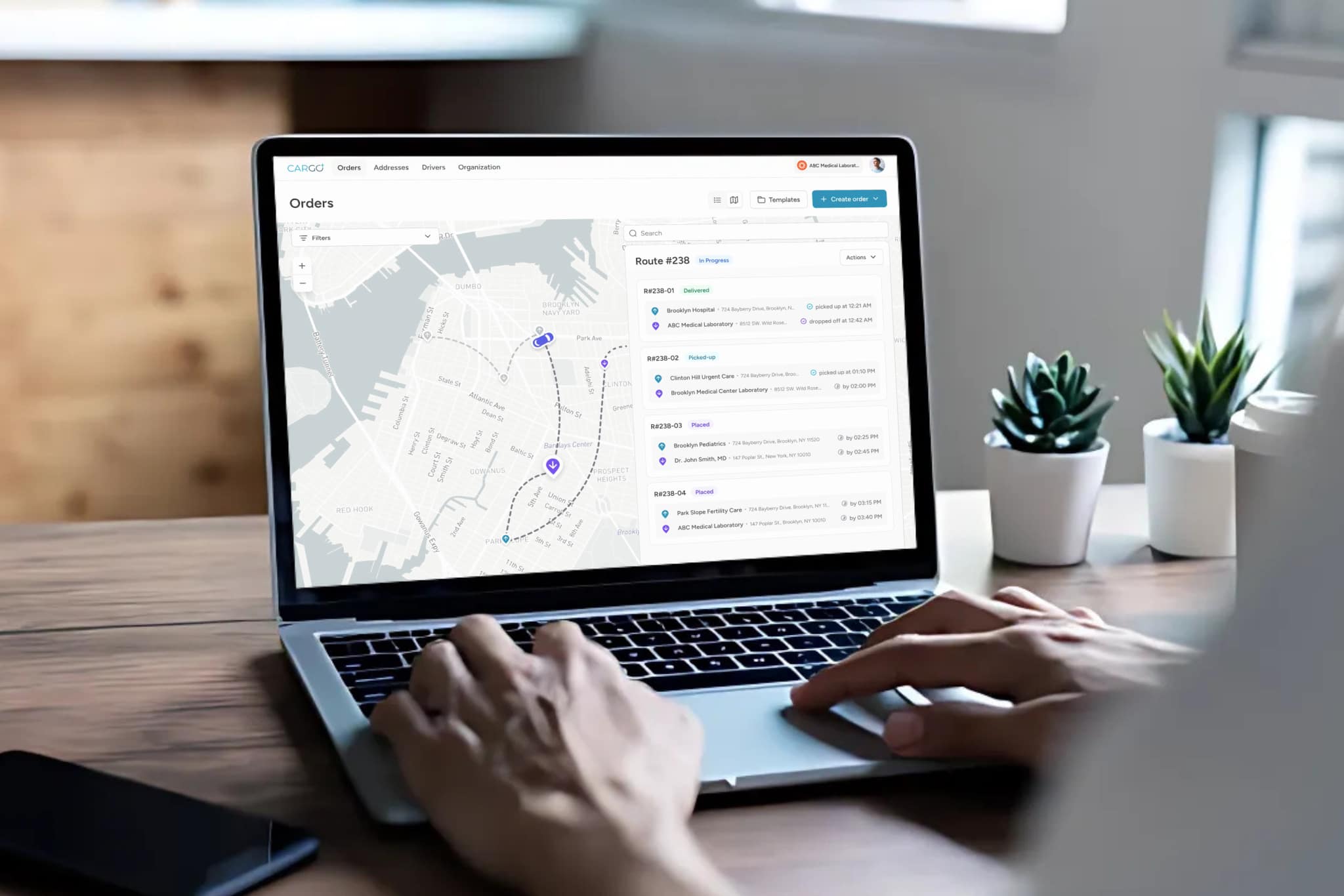
An effective digital chain of custody system integrates multiple technology components into a unified platform. The driver application captures electronic signatures, scans specimen barcodes, records condition notes, and documents photographic evidence at each handoff. The dispatch platform provides real-time visibility into all active specimens, enabling proactive management of exceptions before they become incidents.
Integration with temperature monitoring systems adds environmental data to the chain of custody record, documenting not just who handled the specimen and when, but what temperature conditions it experienced throughout transit. This combined positional and environmental traceability provides the most comprehensive chain of custody documentation available and satisfies the most stringent regulatory requirements.
The analytics layer built on chain of custody data enables continuous improvement. By analyzing handoff times, exception frequencies, route patterns, and temperature compliance across thousands of deliveries, logistics teams can identify systemic issues and implement targeted improvements that reduce error rates and strengthen operational performance over time.
Technology Components:
- Mobile driver application with barcode scanning, electronic signatures, condition notes, and photo documentation
- Centralized dispatch platform providing real-time visibility into all active specimens and couriers
- Integrated temperature monitoring that records environmental conditions as part of the chain of custody record
- Client-facing portal enabling healthcare providers to track specimen status and access delivery documentation
- Analytics engine that identifies patterns, exceptions, and improvement opportunities across all chain of custody data
5. Implementing Chain of Custody Best Practices
Implementing robust chain of custody practices requires alignment between the healthcare provider, the laboratory, and the logistics partner. Standard operating procedures must define exactly what documentation is required at each handoff point, what condition assessments must be performed, and what exception escalation protocols apply. These procedures should be developed collaboratively and reviewed regularly to reflect operational changes and regulatory updates.
Courier training is equally critical. Drivers must understand not just how to use the digital chain of custody tools, but why each documentation step matters. A courier who understands that a missed barcode scan creates a traceability gap that could invalidate a diagnostic result approaches documentation differently than one who views it as an administrative checkbox. This is the caliber of training that separates a purpose-built medical courier from a general delivery provider.
Implementation Best Practices:
- Define chain of custody SOPs collaboratively with laboratory, clinical, and logistics stakeholders
- Train couriers on both the tools and the clinical significance of every documentation step
- Conduct regular compliance audits using chain of custody data to verify adherence to SOPs
- Establish exception escalation protocols that trigger immediate notification and corrective action
- Review and update chain of custody procedures quarterly to reflect regulatory changes and operational improvements
Key Takeaways
Chain of custody in medical specimen transport is the foundation of both regulatory compliance and diagnostic integrity. Digital chain of custody systems provide the traceability, transparency, and auditability that paper-based processes cannot match. For healthcare organizations that rely on outreach specimen logistics, investing in robust chain of custody infrastructure, whether built internally or provided through a specialized logistics partner, is essential to maintaining accreditation, protecting patient outcomes, and building referring provider confidence.
If your organization needs full digital chain of custody with real-time tracking, continuous temperature monitoring, and audit-ready documentation, request a demo to see how carGO Health provides end-to-end specimen traceability across every delivery.
Frequently Asked Questions
What is chain of custody in medical specimen transport?
Chain of custody is the documented, unbroken record of every person and process that handles a specimen from collection to laboratory analysis. It includes timestamped handoffs, electronic signatures, condition assessments, GPS tracking, and environmental monitoring data, providing full traceability for compliance and quality assurance purposes.
Why is chain of custody important for laboratory accreditation?
CLIA and CAP accreditation standards require laboratories to demonstrate control over the entire specimen lifecycle, including the transport phase. During inspections, auditors request documentation showing who handled specimens, when handoffs occurred, and what conditions were maintained. Chain of custody records provide this essential audit documentation.
What is the difference between digital and paper-based chain of custody?
Paper-based systems rely on manual signatures and logs that can be lost, damaged, or incomplete. Digital systems use electronic signatures, barcode scanning, GPS tracking, and automated audit trails to provide real-time visibility and unambiguous documentation at every handoff point, eliminating the traceability gaps common in paper-based processes.
How does chain of custody protect against pre-analytical errors?
Chain of custody systems prevent pre-analytical errors by documenting specimen identity, condition, and environmental exposure at every transition point. Barcode scanning prevents mislabeling, temperature monitoring catches excursions in real time, and automated alerts notify teams of deviations before specimens are compromised.
What technology is needed for digital chain of custody?
Digital chain of custody requires a mobile driver application with barcode scanning and electronic signatures, a centralized dispatch platform with real-time tracking, integrated temperature monitoring, automated exception alerting, and an analytics engine for continuous improvement. A client-facing portal for specimen status visibility is also recommended.