Cold Chain Logistics in Healthcare: Protecting Specimen and Medication Integrity

In healthcare, cold chain logistics refers to the continuous, temperature-controlled supply chain required to transport specimens, pharmaceuticals, biologics, and other sensitive materials from origin to destination without interruption. A single break in the cold chain can render a specimen diagnostically useless, degrade a medication to the point of inefficacy, or compromise a biologic product worth thousands of dollars. For healthcare organizations managing high-volume temperature-controlled transport, cold chain integrity is not a logistics preference. It is a clinical requirement.
The complexity of cold chain logistics in healthcare extends far beyond keeping things cold. Different materials require different temperature ranges, different packaging protocols, and different monitoring standards. A coagulation panel requires refrigeration at 2-8 degrees Celsius, while a frozen tissue biopsy requires dry ice at -78.5 degrees. An mRNA-based therapeutic may require ultra-cold storage below -60 degrees. Managing these requirements simultaneously across dozens of daily routes demands purpose-built infrastructure, trained couriers, and real-time environmental monitoring throughout the entire transport chain.

1. Why Cold Chain Integrity Matters in Healthcare
Cold chain failures in healthcare produce consequences that extend far beyond wasted product. When a specimen degrades during transport due to temperature excursion, the result is often a pre-analytical error that compromises diagnostic accuracy. When a medication loses potency because of improper temperature management, patient safety is directly at risk. When a biologic product worth $10,000 or more is rendered unusable, the financial impact is immediate and significant.
According to the World Health Organization, up to 25 percent of vaccines and temperature-sensitive medications reach their destination in a degraded state due to cold chain failures. In the clinical laboratory space, temperature-related specimen rejection is one of the leading causes of delivery errors that drive up costs and delay patient care. These failures are preventable with the right logistics infrastructure.
Clinical Consequences of Cold Chain Failure:
- Specimen degradation leading to inaccurate diagnostic results or required recollection
- Medication potency loss resulting in therapeutic failure or adverse patient outcomes
- Biologic product waste, including monoclonal antibodies, cell therapies, and vaccines
- Regulatory violations under CLIA, FDA, and state pharmacy board requirements
- Loss of accreditation, client contracts, and institutional reputation
2. Temperature Tiers in Medical Transport
Healthcare cold chain logistics operates across five distinct temperature tiers, each with specific packaging, monitoring, and handling requirements. A medical courier that cannot support all five tiers forces healthcare organizations to use multiple vendors, introducing handoff risk and coordination complexity that increases the probability of cold chain failure.
Understanding the material requirements within each tier is essential for both healthcare providers and their logistics partners. A courier that treats all “cold” shipments identically, without distinguishing between refrigerated transport and cryogenic transport, is a cold chain liability waiting to create a clinical incident.
The Five Temperature Tiers:
- Ambient (15-25 degrees Celsius): routine chemistry panels, hematology specimens, urinalysis, many oral medications
- Refrigerated (2-8 degrees Celsius): coagulation studies, immunoassays, insulin, certain biologics, many vaccines
- Frozen (-20 degrees Celsius): drug level testing, specialized hormone panels, reference laboratory specimens
- Dry ice (-78.5 degrees Celsius): molecular diagnostics, PCR-based testing, tissue specimens for genetic analysis, certain cell therapies
- Cryogenic (-150 degrees Celsius and below): reproductive specimens, stem cells, cord blood, certain research materials
3. Continuous Temperature Monitoring and Real-Time Alerts
Packing a specimen in an insulated container with a cold pack is not cold chain management. It is hope-based logistics. True cold chain integrity requires continuous temperature monitoring from the moment a material is picked up to the moment it is delivered and accessioned, with real-time tracking and transparency at every step.
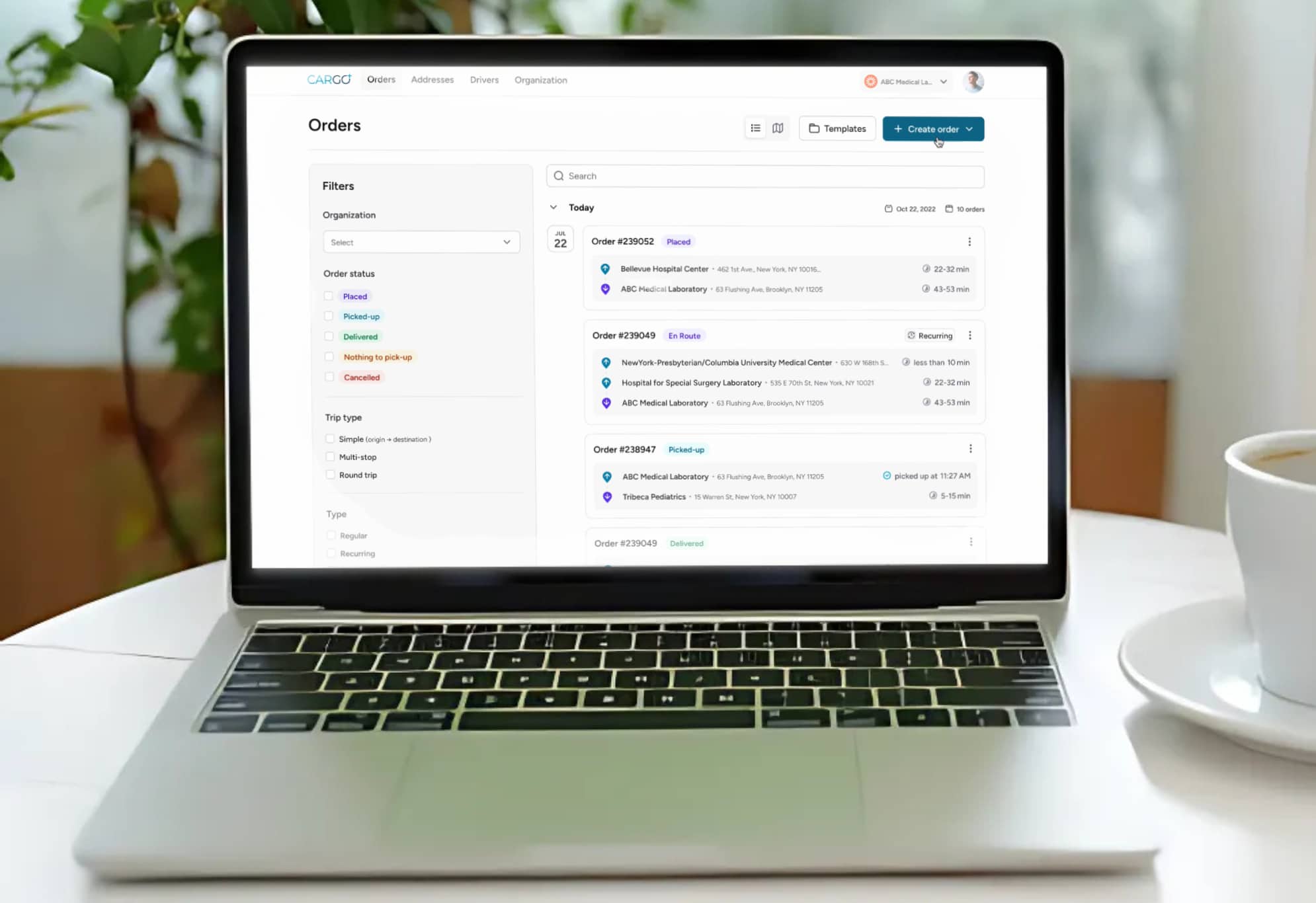
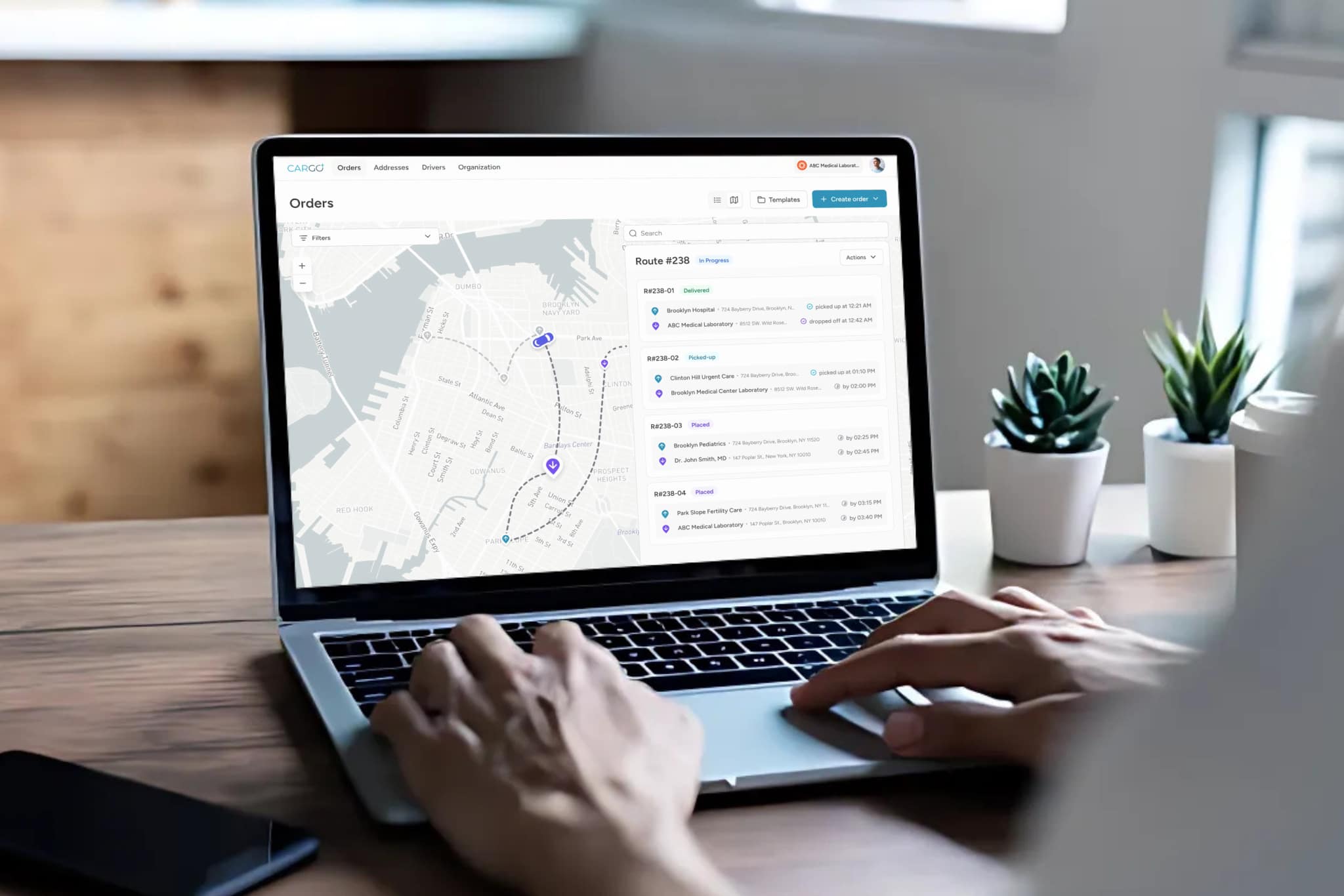
Modern temperature monitoring systems use calibrated digital data loggers that record temperature readings at regular intervals throughout transit. When integrated with a logistics management platform, these systems can generate automated alerts when temperatures deviate from acceptable ranges, enabling couriers and dispatch teams to take corrective action before materials are compromised. This is the difference between reactive incident reporting and proactive cold chain management.
For healthcare providers evaluating their logistics partners, the critical question is not whether the courier uses insulated packaging. It is whether the courier provides documented, continuous temperature data for every shipment, with real-time exception alerting and auditable records that satisfy regulatory inspection requirements.
Effective Temperature Monitoring Includes:
- Calibrated digital data loggers recording temperatures at defined intervals throughout the entire transport duration
- Automated threshold-based alerts sent to dispatch and receiving facility teams when excursions are detected
- Downloadable temperature logs that serve as documentation for CLIA, CAP, FDA, and state regulatory audits
- Integration with GPS tracking to correlate temperature events with location, route, and environmental data
- Historical analytics that identify recurring temperature risks by route, time of day, or seasonal pattern
4. Packaging Validation and Cold Chain Protocols
Temperature monitoring tells you what happened. Validated packaging protocols determine what will happen. Every cold chain shipment should use packaging that has been validated for the specific temperature range, expected transit duration, and ambient environmental conditions of the route. A packaging configuration validated for a 60-minute refrigerated delivery in January may fail during a 90-minute delivery in August.
Packaging validation involves testing insulated containers, cold packs, dry ice quantities, and phase-change materials under simulated conditions to confirm they maintain the target temperature range for the expected transit window plus a safety margin. For biologics and cold chain transport, this validation is not optional. FDA regulations and manufacturer specifications require documented evidence that packaging systems perform as intended.
Cold Chain Packaging Best Practices:
- Use packaging systems validated for the specific temperature tier, transit duration, and seasonal conditions
- Match cold pack or dry ice quantities to the material volume and expected transit time plus a 30 percent safety margin
- Pre-condition packaging materials according to manufacturer specifications before loading specimens or medications
- Document and standardize packaging protocols by shipment type so every courier follows the same validated procedure
- Conduct quarterly packaging performance reviews using temperature log data to identify and address any degradation
5. Building a Resilient Cold Chain Operation
A resilient cold chain operation integrates trained personnel, validated processes, real-time technology, and continuous improvement into a single system. It starts with courier training that goes beyond basic delivery procedures to include specimen science, temperature management, and regulatory awareness. A courier who does not understand why a coagulation specimen must remain at 2-8 degrees is a courier who will eventually mishandle one.
Dispatch and routing must account for temperature sensitivity, not just geography. AI-powered dispatch systems that factor in specimen stability windows, ambient temperature conditions, and real-time traffic data can route time-critical cold chain shipments more effectively than static route planning. This is the kind of operational intelligence that separates a purpose-built medical courier from a general logistics provider attempting to handle healthcare materials.
For healthcare organizations managing complex cold chain requirements across multiple facilities, the logistics partner selection decision is one of the most consequential operational choices they can make. The right partner provides the infrastructure, technology, and expertise to protect cold chain integrity at scale. The wrong partner introduces risk that compounds with every shipment.
Building Cold Chain Resilience:
- Invest in specialized courier training that covers temperature science, packaging protocols, and material-specific handling requirements
- Deploy AI-powered dispatch that factors temperature sensitivity and stability windows into routing decisions
- Implement continuous temperature monitoring with automated alerts across all temperature tiers
- Establish quarterly performance reviews analyzing temperature compliance, excursion rates, and corrective actions
- Partner with a medical courier that supports all five temperature tiers to eliminate multi-vendor cold chain gaps
Key Takeaways
Cold chain logistics in healthcare is not a single capability. It is a system of interconnected disciplines, from validated packaging and continuous monitoring to trained personnel and intelligent routing, that must function together without interruption. Every break in the chain introduces risk to diagnostic accuracy, medication efficacy, patient safety, and regulatory compliance. For healthcare organizations that transport temperature-sensitive materials, building or partnering with a cold chain operation that meets clinical-grade standards is not a logistics upgrade. It is a patient safety imperative.
If your organization needs a logistics partner that supports all five temperature tiers with continuous monitoring and trained medical couriers, request a demo to see how carGO Health protects cold chain integrity across every shipment.
Frequently Asked Questions
What is cold chain logistics in healthcare?
Cold chain logistics in healthcare is the continuous, temperature-controlled supply chain used to transport specimens, medications, biologics, and other sensitive materials from pickup to delivery. It requires validated packaging, continuous temperature monitoring, trained couriers, and real-time alerting to maintain material integrity throughout transit.
What temperature ranges are required for medical specimen transport?
Medical specimens require five temperature tiers: ambient (15-25 degrees Celsius) for routine chemistry, refrigerated (2-8 degrees) for coagulation studies, frozen (-20 degrees) for drug levels, dry ice (-78.5 degrees) for molecular diagnostics, and cryogenic (-150 degrees and below) for reproductive specimens and stem cells.
How do cold chain failures affect patient care?
Cold chain failures can cause specimen degradation leading to inaccurate diagnostic results, medication potency loss resulting in therapeutic failure, and biologic product waste. These failures delay diagnosis, compromise treatment effectiveness, and can result in patient harm. They also trigger regulatory consequences under CLIA, FDA, and state pharmacy board requirements.
What should healthcare providers look for in a cold chain logistics partner?
Healthcare providers should evaluate cold chain partners based on their ability to support all five temperature tiers, provide continuous temperature monitoring with real-time alerts, deliver downloadable temperature logs for regulatory audits, use validated packaging protocols, and employ couriers trained in specimen science and temperature management.
How does temperature monitoring work during medical transport?
Continuous temperature monitoring uses calibrated digital data loggers that record temperatures at defined intervals throughout transit. These loggers integrate with logistics platforms to generate automated alerts when temperatures deviate from acceptable ranges, enabling corrective action before materials are compromised. Temperature data is stored for regulatory audit documentation.