Pre-Analytical Errors: How Specimen Handling Impacts Diagnostic Accuracy
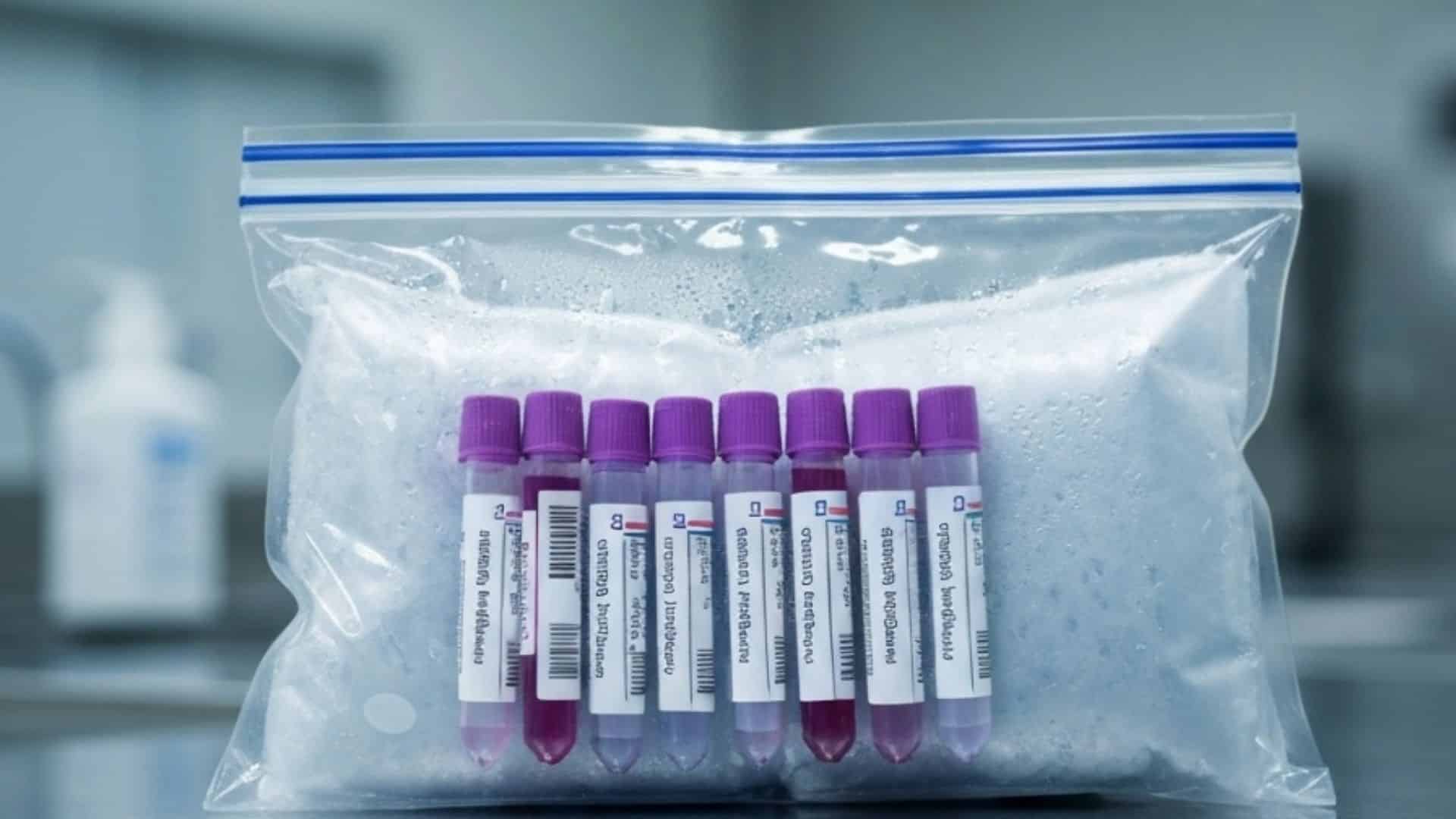
In clinical diagnostics, accuracy depends on far more than the testing instrument or the technologist running it. Research consistently shows that up to 70% of all laboratory errors are pre-analytical errors, failures that occur between the moment a specimen is collected and the moment it reaches the analyzer. These errors include improper temperature exposure, excessive transit times, rough handling that causes hemolysis, mislabeling, and chain-of-custody gaps. Each one has the potential to compromise specimen integrity and produce unreliable diagnostic results.
For laboratories, hospitals, and healthcare networks that rely on outreach specimen collection, the pre-analytical phase introduces the highest concentration of risk in the entire testing workflow. Understanding where these failures originate and how to prevent them is essential to protecting diagnostic accuracy, reducing operational costs, and maintaining CLIA and CAP compliance.

1. What Are Pre-Analytical Errors and Why Do They Matter?
Pre-analytical errors encompass any failure that occurs between specimen collection and laboratory analysis. This phase includes collection technique, labeling, packaging, environmental control during transport, and intake processing at the receiving facility. Because these steps involve multiple handoff points, environmental variables, and human interactions, they represent the most error-prone segment of the diagnostic process.
The consequences of pre-analytical errors extend well beyond a single rejected sample. Compromised specimens can produce results that appear valid but are clinically inaccurate, leading to misdiagnosis, inappropriate treatment plans, and delayed interventions. According to published laboratory quality studies, pre-analytical failures account for the majority of diagnostic errors that directly affect patient outcomes.
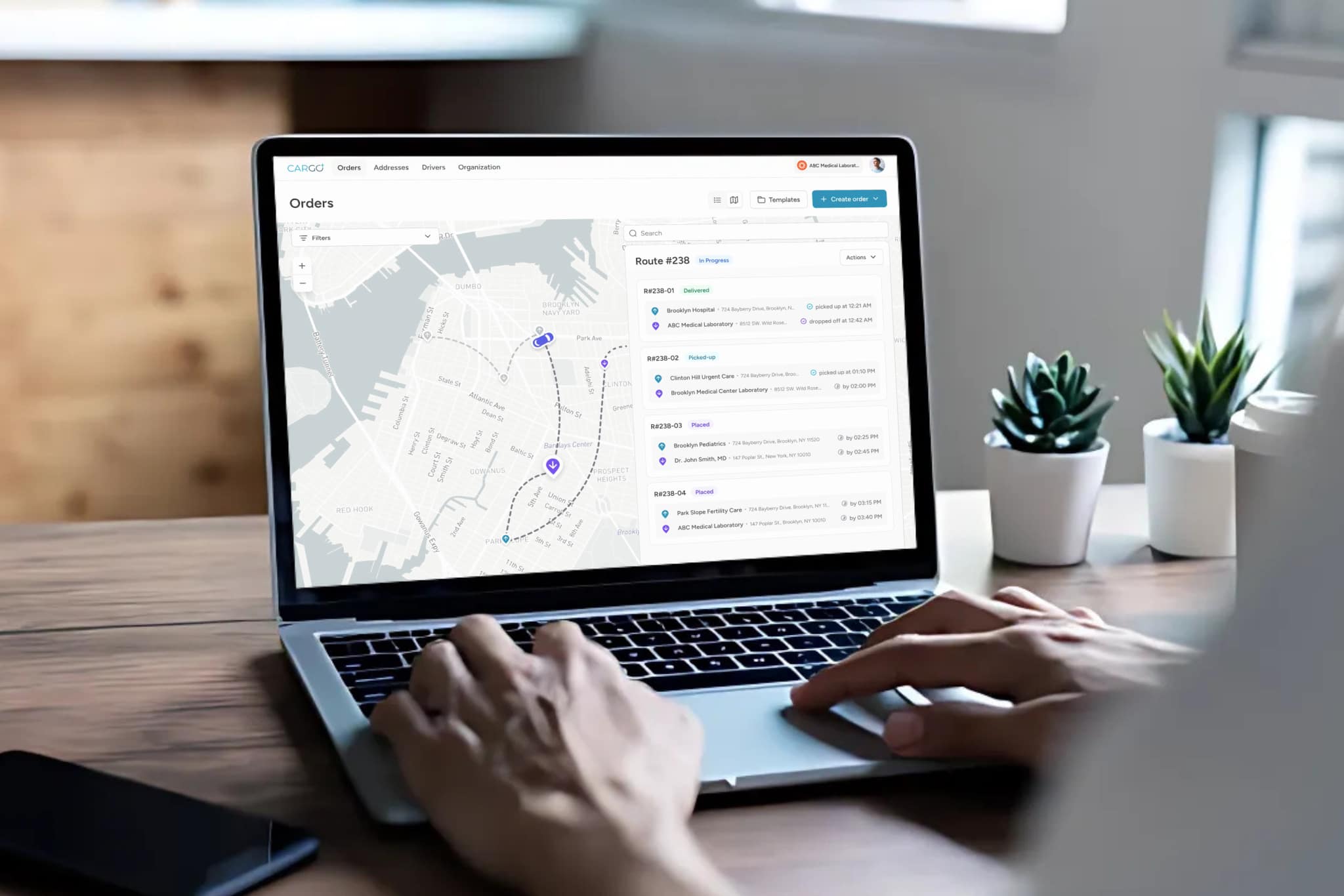
In outreach settings, where specimens must travel from clinics, hospitals, or patient homes to centralized laboratories, the risk is amplified. Unlike in-house workflows where a specimen moves within a controlled environment, outreach logistics expose samples to temperature fluctuations, vibration, prolonged transit, and chain-of-custody gaps that would never occur within a single facility.
Common Types of Pre-Analytical Errors:
- Hemolysis caused by excessive agitation, temperature shock, or improper handling during transport
- Specimen degradation from exposure outside required temperature ranges
- Mislabeling or misidentification during handoff between collection site and courier
- Delayed processing caused by inefficient routing or missed pickup windows
- Loss of chain-of-custody documentation during multi-stop routes
- Clotted samples resulting from inadequate mixing or prolonged pre-transport hold times
2. The Financial and Clinical Cost of Specimen Rejection
When a specimen arrives at a laboratory in a compromised state, the consequences cascade across both clinical and financial dimensions. Degraded specimens can produce inaccurate results that go undetected, leading to misdiagnosis, inappropriate treatment decisions, and delayed interventions. In oncology, a thermally degraded tissue biopsy may yield false-negative results, postponing critical treatment. In cardiology, a hemolyzed blood specimen can falsely elevate potassium levels, triggering unnecessary interventions.
Specimen rejection rates represent a significant and often underestimated cost of delivery errors in healthcare. Each rejected specimen requires patient recall, recollection, and repeat transport. For laboratories, high rejection rates erode client confidence, threaten outreach contracts, and create compliance exposure during accreditation audits.
Direct Costs of Specimen Failure:
- Recollection and retesting expenses, often ranging from $20 to $200+ per specimen depending on test complexity
- Overtime and rescheduling costs for clinical and phlebotomy teams
- Patient dissatisfaction and increased no-show rates for repeat appointments
- Regulatory fines and corrective action requirements from CLIA, CAP, or state health department audits
- Loss of referring provider relationships and outreach revenue due to perceived quality issues
3. Temperature Control: The Most Critical Variable in Specimen Integrity
Temperature is the single most influential environmental factor in specimen integrity. Different specimen types require specific temperature-controlled transport conditions, from ambient handling for routine chemistry panels to cryogenic transport for certain genetic and molecular specimens. Even brief exposure outside the required range can trigger irreversible cellular changes that compromise analytical validity.
Coagulation studies, for example, are highly sensitive to temperature. Specimens held above 24 degrees Celsius begin to show factor degradation within 30 minutes, producing falsely prolonged PT and aPTT results. Ammonia levels rise rapidly in specimens that are not immediately chilled. Lactic acid concentrations shift within minutes at room temperature, potentially leading clinicians to miss or overdiagnose metabolic acidosis.
Effective temperature management requires more than insulated packaging. It demands continuous monitoring throughout the entire transport chain, with real-time tracking and alerting when conditions deviate from acceptable parameters. This level of visibility allows both courier teams and receiving laboratories to make informed decisions about specimen viability before processing begins.
Temperature Management Requirements by Specimen Type:
- Ambient (15-25 degrees Celsius): routine chemistry, hematology, urinalysis
- Refrigerated (2-8 degrees Celsius): coagulation studies, many immunoassays, arterial blood gases
- Frozen (-20 degrees Celsius): drug levels, certain hormone panels, specialized reference lab testing
- Dry ice (-78.5 degrees Celsius): molecular diagnostics, PCR-based testing, tissue specimens for genetic analysis
- Cryogenic (-150 degrees Celsius and below): reproductive specimens, stem cells, certain research materials
4. Chain of Custody and Specimen Traceability
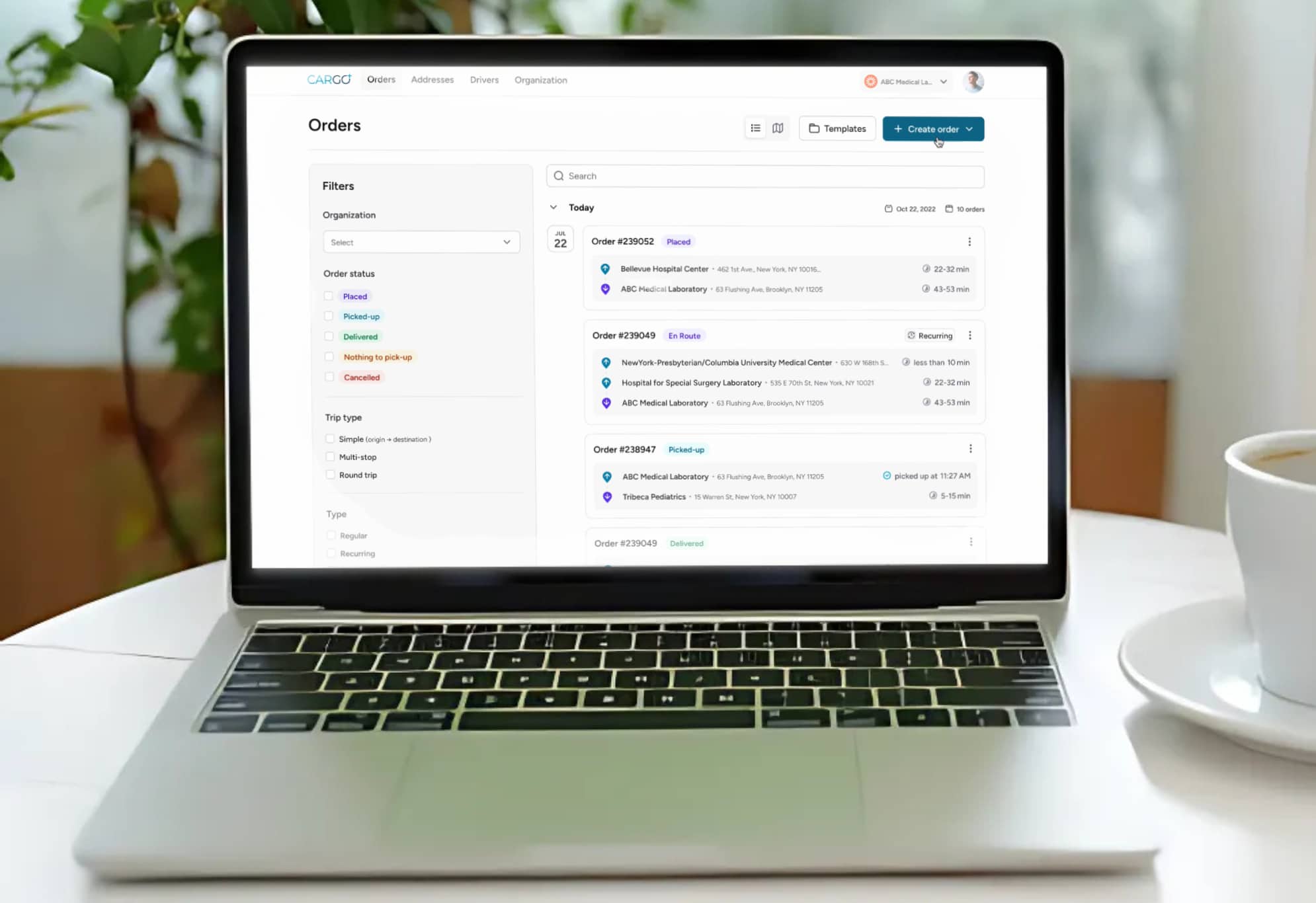
A robust chain-of-custody framework serves two functions: regulatory compliance and error prevention. Every specimen should be tracked from the moment it is collected to the moment it is accessioned at the laboratory, with documented handoffs, timestamps, and condition notes at each transition point. Gaps in this chain create opportunities for misrouting, contamination, and loss of accountability.
Digital chain-of-custody systems eliminate the ambiguity of paper-based tracking. They provide an auditable record that supports HIPAA compliance, satisfies CLIA and CAP accreditation requirements, and gives healthcare providers full visibility into the status of every active specimen in transit. When integrated with real-time GPS tracking, these systems can also alert logistics teams to route deviations, unexpected stops, or delayed handoffs before they result in specimen compromise.
For laboratories managing outreach programs, digital traceability is not a luxury. It is a baseline requirement for maintaining accreditation, defending against audit findings, and demonstrating due diligence in the event of a disputed result. Every untracked handoff is a potential point of failure that can cascade into clinical consequences.
Essential Chain-of-Custody Components:
- Digital documentation of every pickup, handoff, and delivery with timestamps and electronic signatures
- Barcode or QR-based specimen identification to eliminate manual labeling errors at each transition point
- Real-time GPS tracking that provides both courier position and estimated delivery time
- Automated audit trails that support CLIA, CAP, and state regulatory inspections
- Exception alerting for route deviations, temperature excursions, or missed time windows
5. Building a Pre-Analytical Error Prevention Strategy
Reducing pre-analytical errors requires a systems-level approach that addresses people, process, and technology simultaneously. Courier training must extend beyond basic navigation and pickup procedures to include specimen handling science, temperature management protocols, and regulatory awareness. Generic delivery drivers who transport packages during the day and specimens at night do not have the specialized knowledge required to protect diagnostic integrity.
Dispatch and routing systems must account for specimen stability windows, not just geographic efficiency. A STAT specimen with a 60-minute stability window cannot be batched with routine pickups across a multi-stop route. Intelligent routing that prioritizes time-sensitive specimens and dynamically adjusts based on real-time conditions is essential to preventing pre-analytical failures at scale.
Healthcare providers should evaluate their logistics partners based on pre-analytical performance metrics, including specimen rejection rates, temperature compliance records, on-time pickup performance, and chain-of-custody completeness. These metrics provide an objective foundation for continuous improvement and accountability. The difference between a purpose-built medical courier and a general logistics provider is not a matter of convenience. It is a matter of diagnostic integrity.
Strategic Prevention Measures:
- Specialized courier certification in specimen science, handling protocols, and regulatory compliance (HIPAA, OSHA, DOT)
- AI-powered dispatch systems that optimize routes based on specimen priority, stability windows, and real-time traffic conditions
- Continuous temperature monitoring with automated threshold alerts throughout the entire transport chain
- Integrated analytics platforms that track rejection rates, transit times, and temperature compliance across all routes
- Collaborative quality review processes between courier providers and laboratory quality assurance teams
Key Takeaways
Pre-analytical errors represent one of the most preventable sources of diagnostic failure in healthcare. By investing in purpose-built logistics infrastructure, including temperature-controlled transport, digital chain of custody, real-time monitoring, and specialized courier training, healthcare organizations can dramatically reduce specimen rejection rates, improve diagnostic accuracy, and protect patient outcomes. For any organization that depends on outreach specimen logistics, eliminating pre-analytical errors is not an operational improvement. It is a clinical imperative.
If your organization is evaluating how to reduce specimen rejection rates and strengthen pre-analytical quality, request a demo to see how purpose-built medical courier infrastructure can protect diagnostic integrity across your entire specimen network.
Frequently Asked Questions
What is the most common pre-analytical error in specimen handling?
Hemolysis is the most frequently reported pre-analytical error, often caused by improper handling, excessive agitation, or temperature shock during specimen transport. Hemolyzed samples affect potassium, LDH, AST, and other analyte measurements, leading to inaccurate results that may require recollection.
How do pre-analytical errors affect diagnostic accuracy?
Pre-analytical errors can produce false-positive or false-negative results, delay diagnosis, and lead to inappropriate treatment decisions. Up to 70% of all laboratory errors originate in the pre-analytical phase, making it the single largest source of diagnostic inaccuracy in clinical testing workflows.
What temperature requirements apply to medical specimen transport?
Temperature requirements vary by specimen type. Routine chemistry specimens require ambient conditions (15-25 degrees Celsius), coagulation studies need refrigeration (2-8 degrees Celsius), molecular diagnostics often require dry ice (-78.5 degrees Celsius), and reproductive or stem cell specimens may require cryogenic transport (-150 degrees Celsius or below).
How can laboratories reduce specimen rejection rates?
Laboratories can reduce rejection rates by partnering with specialized medical couriers that provide continuous temperature monitoring, digital chain of custody, AI-optimized routing that accounts for specimen stability windows, and trained couriers who understand specimen handling science. Tracking rejection rate data by route, courier, and specimen type helps identify and address recurring failure points.
What is chain of custody in medical specimen transport?
Chain of custody is the documented record of every person and process that handles a specimen from collection to laboratory analysis. It includes timestamped pickups, handoffs, and deliveries with electronic signatures, condition notes, and GPS data. Digital chain-of-custody systems support HIPAA compliance, CLIA accreditation, and provide full traceability for audit purposes.