OSHA and DOT Compliance for Medical Couriers: What Healthcare Providers Must Know

Medical courier operations are governed by a regulatory framework that most general delivery services never encounter. OSHA (Occupational Safety and Health Administration) and DOT (Department of Transportation) regulations impose specific requirements on how medical materials are handled, packaged, labeled, and transported. These are not optional best practices. They are federal mandates that carry significant penalties for non-compliance and, more importantly, exist to protect couriers, healthcare workers, and patients from occupational hazards and transport-related incidents involving biological substances and hazardous materials.
For healthcare organizations that rely on courier services to transport specimens, pharmaceuticals, and medical devices, understanding these compliance requirements is essential. Partnering with a courier that does not meet OSHA and DOT standards does not transfer the compliance burden. It amplifies the liability. An OSHA-compliant medical courier is not a premium service tier. It is the baseline requirement for any organization transporting regulated healthcare materials.

1. OSHA Bloodborne Pathogen Standard and Medical Transport
OSHA’s Bloodborne Pathogen Standard (29 CFR 1910.1030) is the primary federal regulation governing occupational exposure to blood and other potentially infectious materials. For medical couriers, this standard applies directly to every pickup, transport, and delivery involving clinical specimens, blood products, and biological materials. Compliance requires a comprehensive exposure control plan, documented training, appropriate personal protective equipment, and post-exposure incident protocols.
Couriers who handle medical specimens must be trained to recognize exposure risks, use PPE correctly, and follow decontamination procedures. They must understand proper specimen packaging, secondary containment requirements, and the procedures for handling a specimen leak or container failure during transport. An untrained courier handling a leaking specimen tube is not just a logistics failure. It is an occupational safety incident with legal and regulatory consequences.
Healthcare organizations that use couriers for specimen transport are responsible under OSHA for ensuring that their logistics partners maintain compliant bloodborne pathogen programs. This responsibility does not transfer to the courier by default. It must be verified through documented training records, exposure control plans, and compliance audits.
OSHA Bloodborne Pathogen Requirements for Couriers:
- Written exposure control plan identifying tasks with exposure risk and mitigation procedures
- Annual bloodborne pathogen training with documented completion for every courier
- Provision and proper use of PPE including gloves, eye protection, and spill response materials
- Hepatitis B vaccination offered to all couriers with occupational exposure potential
- Post-exposure evaluation and follow-up procedures including incident documentation
2. DOT Hazardous Materials Regulations for Medical Shipments
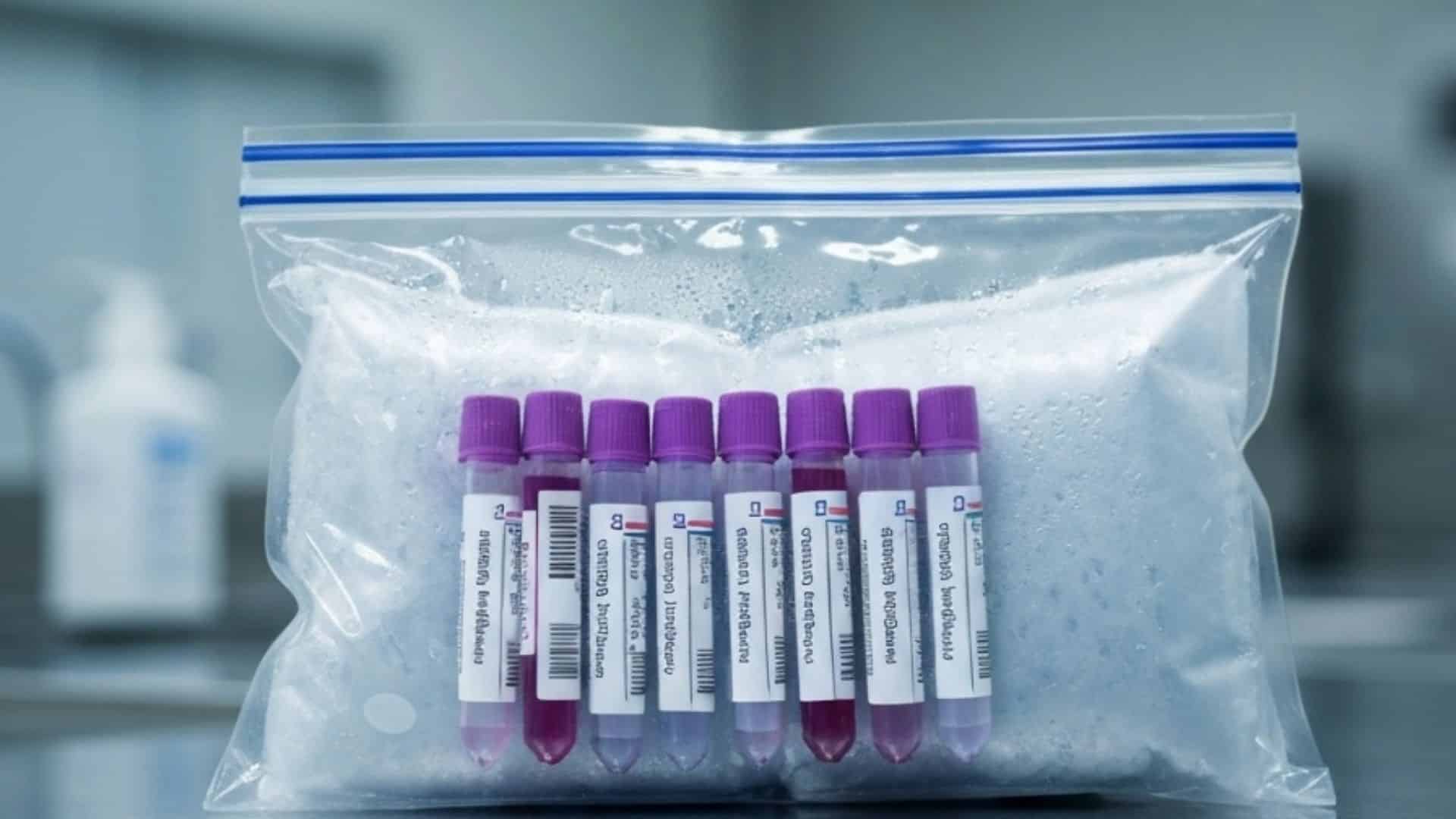
The Department of Transportation regulates the transport of hazardous materials under 49 CFR Parts 171-180. Medical specimens classified as Category B biological substances (UN 3373) and shipments requiring dry ice (UN 1845) fall under these regulations. Compliance requires proper hazmat classification, specific packaging configurations, correct labeling and marking, and driver certification in hazardous materials handling.
Category B biological substances, which include most clinical diagnostic specimens, must be packaged in triple-containment systems: a primary leak-proof container, absorbent material, a secondary leak-proof container, and a rigid outer packaging marked with the UN 3373 diamond. Dry ice shipments carry additional requirements including proper ventilation markings, weight documentation, and packaging designed to allow gas release without pressure buildup.
DOT violations can result in fines up to $75,000 per occurrence, and criminal penalties for willful violations can reach $500,000 with potential imprisonment. For medical courier operations handling hundreds or thousands of regulated shipments per week, a systemic compliance failure can create exposure measured in millions of dollars.
DOT Compliance Requirements:
- Proper hazmat classification of all transported materials (Category A, Category B, exempt specimens)
- Triple-containment packaging systems meeting UN 3373 specifications for Category B substances
- Correct labeling and marking on all outer packaging per 49 CFR requirements
- Hazmat training and certification for all drivers handling regulated materials, renewed every three years
- Dry ice quantity documentation, ventilation markings, and packaging that permits gas release
3. HIPAA Compliance in Medical Transport
While OSHA and DOT address physical safety, HIPAA compliance addresses information security during medical transport. Medical couriers regularly handle specimens accompanied by requisition forms, patient labels, and other materials containing protected health information (PHI). Under HIPAA, couriers may function as business associates of covered entities, creating specific obligations for PHI protection during transport.
HIPAA compliance for medical couriers extends beyond simply not reading patient labels. It encompasses secure handling of all materials containing PHI, access controls that restrict information to authorized personnel, incident response procedures for potential breaches, and business associate agreements that document the courier’s obligations under the HIPAA Privacy and Security Rules.
A HIPAA-compliant courier operation implements these protections systematically: driver training on PHI handling, secure vehicle storage, digital chain of custody systems that restrict data access, and incident response protocols that satisfy HIPAA breach notification requirements.
HIPAA Transport Compliance:
- Business associate agreements between healthcare organizations and courier providers
- Driver training on PHI recognition, handling requirements, and prohibited disclosure
- Secure vehicle storage that prevents unauthorized access to materials containing patient information
- Digital systems with access controls that restrict PHI visibility to authorized personnel only
- Breach notification procedures that satisfy HIPAA’s 60-day reporting requirement
4. State-Specific Regulatory Requirements
Beyond federal OSHA, DOT, and HIPAA regulations, medical courier operations must also comply with state-specific requirements that vary significantly across jurisdictions. States may impose additional licensing requirements, vehicle inspection standards, insurance minimums, and operational permits for medical transport services. For couriers operating across multiple states, as is common in the Northeast corridor, managing compliance across multiple jurisdictions adds another layer of complexity.
In New York, New Jersey, and Connecticut, for example, pharmacy delivery regulations, clinical laboratory transport requirements, and medical waste handling standards each have state-level specifications that go beyond federal minimums. A medical courier operating across these states must maintain awareness of and compliance with each jurisdiction’s unique requirements.
Common State-Level Requirements:
- State-specific courier or medical transport licensing and registration
- Vehicle inspection and insurance minimums that may exceed federal standards
- Pharmacy board regulations for prescription and controlled substance delivery
- State health department requirements for clinical laboratory specimen transport
- Medical waste generator and transporter permits where applicable
5. Building a Compliance-First Medical Courier Operation
A compliance-first approach means building OSHA, DOT, HIPAA, and state regulatory requirements into the foundation of operations rather than adding them as an afterthought. This starts with hiring practices that screen for candidates capable of handling regulated materials, continues through comprehensive training programs that cover all applicable frameworks, and is sustained through ongoing compliance monitoring, documentation, and continuous improvement.
For healthcare organizations evaluating courier partners, compliance verification should be a non-negotiable part of the vendor selection process. Request documented evidence of OSHA training records, DOT hazmat certifications, HIPAA policies, and state-specific licensing. Verify that the courier maintains current compliance, not just initial certification. And include compliance performance metrics, such as incident rates, audit findings, and corrective action completion, in ongoing partner evaluations.
The cost of compliance failure in medical courier operations, measured in fines, litigation, accreditation risk, and patient safety exposure, far exceeds the cost of operating a fully compliant logistics program. A purpose-built medical courier treats compliance as a core operational competency, not an expense to be minimized.
Compliance Program Essentials:
- Comprehensive initial training covering OSHA, DOT, HIPAA, and state-specific requirements for every new courier
- Annual recertification and refresher training with documented completion records
- Regular compliance audits with documented findings and corrective action tracking
- Incident reporting and response protocols with root cause analysis for every compliance event
- Continuous monitoring of regulatory changes across all applicable federal and state jurisdictions
Key Takeaways
OSHA, DOT, and HIPAA compliance in medical courier operations is not a differentiator. It is a baseline requirement. Healthcare organizations that transport regulated materials are responsible for ensuring their logistics partners meet all applicable federal and state standards. The regulatory exposure from non-compliant courier operations, including OSHA penalties, DOT fines, HIPAA sanctions, and accreditation risk, creates liability that no organization should accept. Partnering with a medical courier that treats compliance as a core operational competency, not an add-on, is the most effective way to protect patients, couriers, and the organization itself.
If you need a fully compliant medical courier partner with OSHA, DOT, and HIPAA certification across every driver and every route, request a demo to see how carGO Health builds compliance into every aspect of its operations.
Frequently Asked Questions
What OSHA regulations apply to medical couriers?
OSHA’s Bloodborne Pathogen Standard (29 CFR 1910.1030) is the primary regulation. It requires medical couriers to have written exposure control plans, annual training, appropriate PPE, Hepatitis B vaccination offers, and post-exposure incident protocols for anyone handling clinical specimens or biological materials.
What DOT certification do medical courier drivers need?
Drivers handling Category B biological substances (UN 3373) or dry ice (UN 1845) need DOT hazardous materials training covering proper classification, packaging, labeling, and emergency response. This certification must be renewed every three years. Violations can result in fines up to $75,000 per occurrence.
How does HIPAA apply to medical specimen transport?
Medical couriers handling specimens with patient information may function as HIPAA business associates. Compliance requires business associate agreements, driver training on PHI handling, secure vehicle storage, access-controlled digital systems, and breach notification procedures satisfying HIPAA’s 60-day reporting requirement.
What is UN 3373 packaging for medical specimens?
UN 3373 is the classification for Category B biological substances, which includes most clinical diagnostic specimens. Packaging requires triple containment: a primary leak-proof container, absorbent material, a secondary leak-proof container, and rigid outer packaging marked with the UN 3373 diamond symbol per DOT specifications.
What happens if a medical courier is not OSHA or DOT compliant?
Non-compliance can result in OSHA penalties up to $15,625 per serious violation, DOT fines up to $75,000 per occurrence, HIPAA sanctions up to $1.5 million per violation category, and potential criminal penalties. Healthcare organizations using non-compliant couriers may share liability for regulatory violations occurring during transport.