Medical Logistics Services: Streamlining Healthcare Supply Chains in 2026

Updated April 2026 — reviewed and refreshed by carGO Health editorial team.
The healthcare industry moves more than just data and prescriptions. Every day, millions of specimens, pharmaceutical products, medical devices, and biological materials travel between hospitals, laboratories, pharmacies, and clinics across the country. The systems responsible for moving these materials are collectively known as medical logistics services, and they represent one of the most critical yet underexamined components of modern healthcare delivery. Unlike general freight or consumer package delivery, medical logistics operates under a unique set of constraints that demand specialized infrastructure, regulatory compliance, and clinical awareness at every stage of the supply chain. When these systems function well, clinicians receive what they need when they need it and patients receive timely diagnoses and treatments. When they break down, the consequences are measured in delayed care, compromised specimens, wasted medications, and regulatory violations that can shut down an entire operation.
The global healthcare logistics market was valued at approximately $115 billion in 2024 and is projected to exceed $170 billion by 2030, according to industry analyses from Grand View Research. That growth is being driven by the expansion of precision medicine, the rise of decentralized clinical trials, the increasing volume of reference laboratory testing, and the growing complexity of pharmaceutical distribution networks. For healthcare organizations navigating this landscape, understanding what separates genuine medical logistics from general logistics is not an academic exercise. It is a strategic and operational imperative that directly affects patient outcomes, regulatory standing, and financial performance.
1. What Makes Medical Logistics Different From General Logistics
General logistics is built around efficiency, cost optimization, and throughput. The primary objective is moving goods from point A to point B at the lowest possible cost per unit. General logistics providers optimize for speed and volume, but the consequences of a delayed or mishandled shipment are typically financial. A late consumer package results in a refund or a customer complaint. A late medical specimen can result in a misdiagnosis, a delayed treatment decision, or a patient who must undergo a second invasive collection procedure.
This fundamental difference in consequence is what separates healthcare logistics from every other logistics vertical. Medical logistics services must account for regulatory compliance under OSHA bloodborne pathogen standards, DOT hazardous materials regulations, HIPAA privacy requirements, and CLIA laboratory accreditation standards simultaneously. A general logistics provider that handles consumer goods has no framework for any of these requirements. A medical courier service must be built around them from the ground up.
Key Differences Between General and Medical Logistics:
- Regulatory environment: medical logistics requires simultaneous compliance with HIPAA, OSHA, DOT, CLIA, and state-specific health department regulations
- Temperature control: medical materials often require specific temperature ranges from pickup to delivery, with continuous monitoring and documentation
- Time sensitivity: many medical shipments have clinically defined stability windows measured in minutes or hours, not days
- Chain of custody: every handoff must be documented with timestamps, signatures, and condition verification for legal and regulatory traceability
- Personnel requirements: couriers must be trained in biohazard handling, specimen science, patient privacy, and emergency response protocols
- Consequence of failure: errors produce clinical harm, regulatory penalties, and legal liability rather than simple financial loss
Organizations that attempt to use general logistics providers for medical materials are effectively treating clinical supply chain functions as commodity services. The result is predictable: higher rejection rates, compliance gaps, and an operational fragility that becomes visible the first time something goes wrong during a STAT delivery situation where every second counts.
2. The Compliance Framework Behind Medical Logistics Services
Compliance in medical logistics is not a checkbox exercise. It is a layered regulatory framework that touches every aspect of operations, from how a courier is trained to how a vehicle is inspected to how patient information is protected during transport. Understanding this framework is essential for any healthcare organization evaluating its logistics infrastructure or selecting a medical logistics courier partner.
HIPAA compliance governs how protected health information is handled during transport. Every specimen label, requisition form, patient document, and electronic communication associated with a medical shipment contains PHI that must be safeguarded. Couriers must be trained as business associates under HIPAA, with documented training, signed business associate agreements, and operational protocols that prevent unauthorized access or disclosure. A logistics provider that does not maintain these protections exposes healthcare organizations to penalties that can reach $1.5 million per violation category under the HHS HIPAA enforcement framework.
OSHA compliance under the Bloodborne Pathogens Standard (29 CFR 1910.1030) requires that couriers handling clinical specimens receive training in exposure control, use appropriate personal protective equipment, and follow documented procedures for spill containment and post-exposure response. OSHA and DOT compliance requirements for medical couriers are extensive and non-negotiable. DOT adds another layer through 49 CFR Part 173, which governs the packaging, labeling, and transport of infectious substances and diagnostic specimens as hazardous materials.
For organizations that serve as reference laboratories or manage multi-site clinical operations, CLIA and CAP accreditation standards extend compliance requirements to the transport phase of the testing process. The Centers for Medicare and Medicaid Services CLIA regulations explicitly require that specimens be transported under conditions that maintain their integrity for accurate analysis. A logistics partner that cannot demonstrate compliance across all of these frameworks simultaneously is a compliance liability, regardless of how efficient their delivery times may appear.
Healthcare organizations should expect their medical logistics partner to provide documented evidence of compliance across every applicable regulatory domain. This includes training and certification records for couriers, vehicle inspection logs, exposure control plans, business associate agreements, and standard operating procedures for every material type transported.
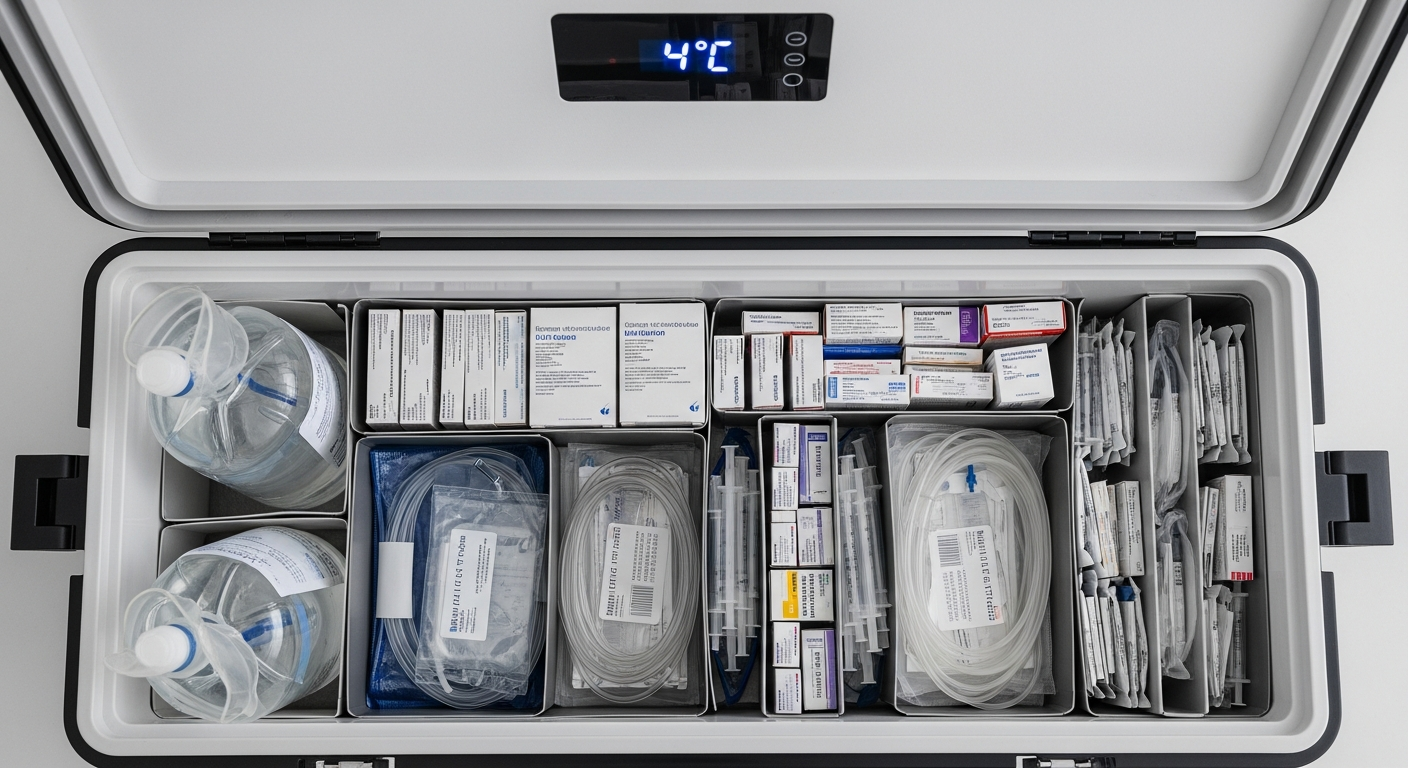
3. Temperature Control and Cold Chain Management in Healthcare Logistics
Temperature control is one of the defining technical challenges of healthcare supply chain logistics. Unlike general freight, where ambient conditions are acceptable for most shipments, medical logistics must manage materials across multiple temperature tiers simultaneously. A single route may include ambient specimens, refrigerated medications, frozen reference laboratory panels, and dry ice shipments for molecular diagnostics, each with its own stability window and packaging protocol.
According to the World Health Organization, temperature-related degradation affects up to 25 percent of vaccines and temperature-sensitive pharmaceuticals during transport globally. In the clinical laboratory space, temperature excursions are a leading cause of pre-analytical errors in specimen handling, which account for up to 70 percent of all laboratory errors according to research published in the National Library of Medicine. These are not abstract statistics. They represent real patients receiving inaccurate results, unnecessary repeat procedures, and delayed treatment decisions.
Modern cold chain logistics in healthcare requires validated packaging systems, continuous temperature monitoring with IoT-enabled data loggers, automated exception alerting, and downloadable temperature records for regulatory audits. The logistics provider must support all five temperature tiers, from ambient through cryogenic, with dedicated packaging protocols and trained couriers who understand the clinical significance of temperature management for each material type.
Temperature Management Requirements in Medical Logistics:
- Validated packaging configurations for each temperature tier, tested for expected transit duration plus a safety margin
- Continuous IoT-enabled temperature monitoring from pickup through delivery with automated threshold alerts
- Courier training on temperature science, including understanding how ambient conditions and transit time affect material stability
- Seasonal packaging adjustments to account for heat exposure in summer and freeze risk in winter
- Auditable temperature records that satisfy CLIA, CAP, FDA, and state regulatory inspection requirements
For healthcare organizations that transport high volumes of temperature-sensitive materials, the choice of logistics partner directly determines specimen rejection rates, medication waste, and regulatory compliance posture. A partner with robust cold chain infrastructure reduces specimen rejection rates and protects the clinical value of every shipment.
4. How Technology Is Transforming Medical Logistics in 2026
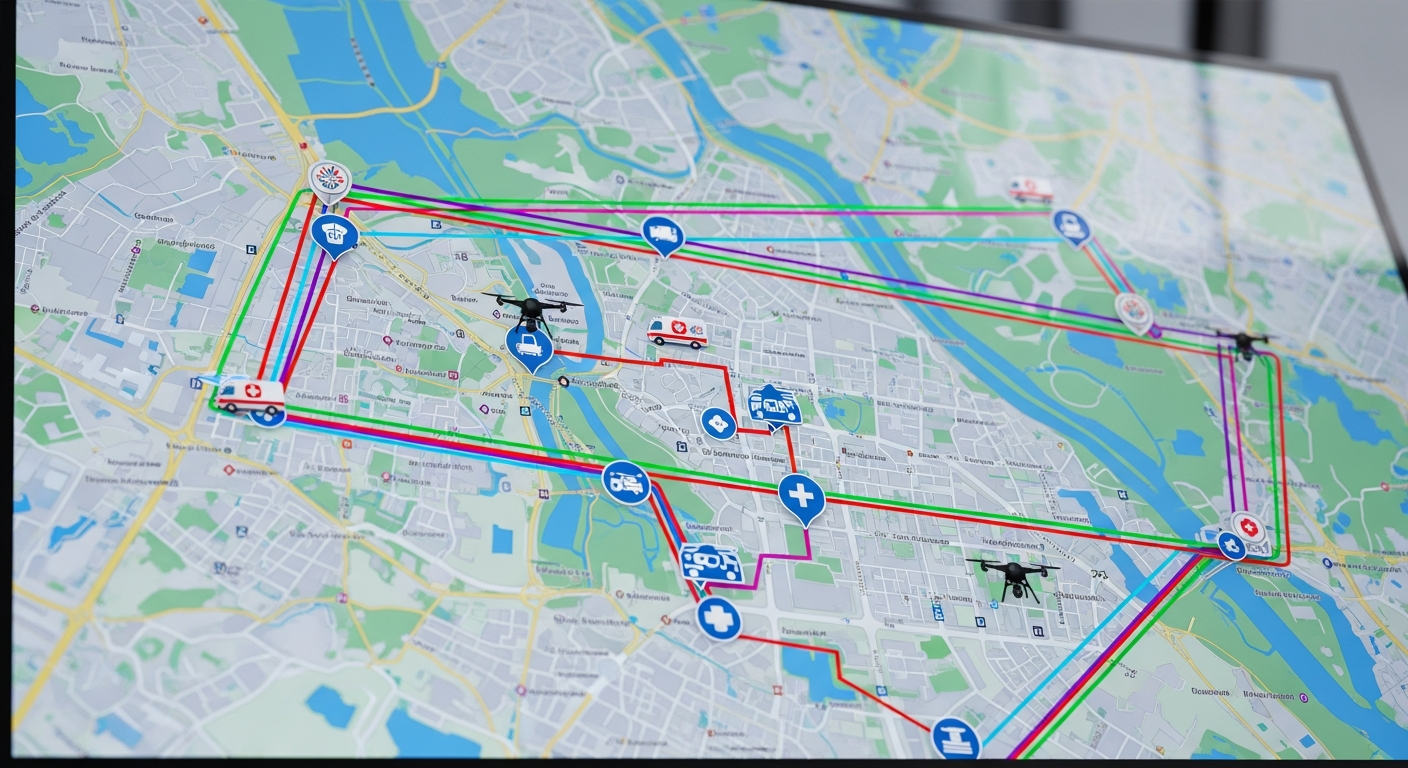
The most significant transformation in medical freight and logistics over the past five years has been the integration of artificial intelligence, real-time data, and digital documentation into every stage of the transport chain. Legacy medical logistics operated on phone calls, paper manifests, and fixed route schedules. Modern medical logistics operates on AI-powered dispatch algorithms, GPS-integrated tracking platforms, digital chain of custody systems, and predictive analytics that optimize operations in real time.
AI-powered dispatch technology represents the most impactful of these advances. Traditional dispatch relies on human dispatchers making routing decisions based on experience, geographic familiarity, and queue order. AI dispatch systems process real-time data on traffic conditions, courier location, specimen stability windows, delivery priority levels, temperature sensitivity, and vehicle capacity to generate optimized routing decisions in seconds. The result is faster delivery times, fewer missed stability windows, lower fuel costs, and more efficient utilization of the courier fleet.
Digital chain of custody has replaced paper-based documentation with electronic systems that capture timestamps, GPS coordinates, condition verification, signature capture, and photographic documentation at every handoff point. This creates an unbroken digital record that satisfies regulatory audit requirements and provides real-time tracking that enhances delivery transparency for sending and receiving facilities. When a laboratory director needs to know where a specimen is at any given moment, the answer is available in real time rather than requiring a phone call to dispatch.
Technology Capabilities in Modern Medical Logistics:
- AI-optimized dispatch that factors in specimen stability windows, traffic patterns, delivery priority, and temperature requirements
- Real-time GPS tracking with estimated arrival times visible to sending and receiving facilities
- Digital chain of custody with electronic signatures, timestamp capture, and photographic condition documentation
- IoT-enabled environmental monitoring for temperature, humidity, and shock detection during transit
- Predictive analytics that identify route inefficiencies, recurring delay patterns, and capacity optimization opportunities
- Automated reporting and compliance documentation generated from transport data without manual data entry
These capabilities are not futuristic concepts. They are operational realities for medical courier organizations that have invested in purpose-built technology platforms. Healthcare organizations evaluating logistics partners should treat technology infrastructure as a core selection criterion, not a secondary consideration. The logistics platform a provider operates on determines the quality of data, visibility, and operational intelligence available to every stakeholder in the supply chain.
5. The End-to-End Medical Logistics Chain: From Pickup to Delivery
Understanding the full lifecycle of a medical logistics transaction reveals why general logistics approaches fail in healthcare. Every stage of the end-to-end chain introduces requirements that do not exist in conventional logistics, and every handoff represents a point where compliance, temperature integrity, or specimen viability can be compromised if proper protocols are not in place.
The end-to-end medical logistics chain includes these critical stages:
Order intake and classification. Every transport request must be classified by material type, temperature requirement, priority level, regulatory category, and special handling instructions. A STAT blood gas specimen requiring ice transport has fundamentally different logistics requirements than a scheduled batch of ambient urinalysis specimens. The intake process must capture these distinctions accurately and route them to the appropriate dispatch workflow.
Dispatch and routing. Once classified, the shipment must be assigned to a qualified courier with the appropriate vehicle, equipment, and training for the material type. Dispatch must account for the specimen stability window, current traffic conditions, other active pickups and deliveries on the route, and the delivery model, whether on-demand or scheduled. This is the stage where AI dispatch delivers the most significant operational improvement over manual routing.
Pickup and verification. At the pickup location, the courier must verify the shipment contents against the manifest, confirm packaging integrity, verify temperature conditions, and initiate chain of custody documentation. For chain of custody in medical specimen transport, this pickup verification is the foundation of legal and regulatory traceability.
In-transit management. During transport, the logistics system must maintain continuous visibility into courier location, estimated arrival time, and environmental conditions. Any exception, whether a traffic delay that threatens a stability window or a temperature excursion detected by an IoT sensor, must trigger an automated response protocol that either resolves the issue or escalates it to the appropriate decision-maker.
Delivery and accessioning. At the destination, the courier must verify the receiving party, confirm material condition, capture delivery confirmation with timestamp and signature, and close the chain of custody record. For laboratory deliveries, this includes specimen accessioning verification to confirm that the right specimens reached the right department in acceptable condition.
Post-delivery documentation and analytics. After delivery, the logistics system should generate a complete transport record including chain of custody documentation, temperature logs, transit time data, and any exception records. This data feeds into compliance reporting, performance analytics, and continuous improvement programs that strengthen the logistics operation over time.
Each of these stages requires specialized capability that general logistics providers do not possess. Healthcare organizations that understand the end-to-end chain can evaluate their logistics partners more effectively and identify the operational gaps that create risk in their supply chain.
Key Takeaways
Medical logistics services are not a subcategory of general logistics. They are a specialized discipline defined by regulatory complexity, clinical consequence, temperature sensitivity, and documentation requirements that do not exist in any other logistics vertical. The healthcare logistics market is growing rapidly as precision medicine, decentralized trials, and multi-site healthcare networks drive increasing volume and complexity. Organizations that treat medical logistics as a commodity service expose themselves to compliance risk, clinical errors, and operational fragility that compounds over time.
The most effective approach to healthcare supply chain logistics is partnering with a purpose-built medical logistics provider that combines regulatory expertise, temperature management infrastructure, trained personnel, and modern technology into an integrated operation. carGO Health delivers this integrated approach with AI-powered dispatch, real-time tracking, certified medical couriers, and full compliance across HIPAA, OSHA, and DOT requirements, backed by over 150,000 completed orders and 100 percent customer retention across the healthcare organizations we serve.
If your organization is evaluating its medical logistics infrastructure or considering a transition from general logistics to a specialized healthcare partner, request a demo to see how carGO Health streamlines the entire supply chain from pickup to delivery.
Frequently Asked Questions
What are medical logistics services?
Medical logistics services encompass the specialized systems, processes, and infrastructure required to transport healthcare materials including specimens, pharmaceuticals, medical devices, and biological products between healthcare facilities. Unlike general logistics, medical logistics requires compliance with HIPAA, OSHA, DOT, and CLIA regulations, temperature-controlled transport across multiple tiers, documented chain of custody, and couriers trained in biohazard handling and specimen science.
How is medical logistics different from general freight logistics?
Medical logistics differs from general freight in several critical ways. It operates under multiple simultaneous regulatory frameworks including HIPAA, OSHA, and DOT. It requires temperature-controlled transport with continuous monitoring. It demands documented chain of custody at every handoff. And the consequences of failure are clinical rather than purely financial, meaning errors can directly affect patient diagnoses, treatment decisions, and safety rather than simply resulting in refunds or delays.
What compliance requirements apply to medical logistics couriers?
Medical logistics couriers must comply with HIPAA requirements for protecting patient health information during transport, OSHA Bloodborne Pathogens Standard for safe handling of clinical specimens, DOT regulations for packaging and labeling infectious substances as hazardous materials, and CLIA standards for maintaining specimen integrity during transport. Couriers must maintain current training certifications, signed business associate agreements, and documented standard operating procedures for each material type.
How does AI technology improve medical logistics operations?
AI technology improves medical logistics by optimizing dispatch and routing decisions in real time. AI-powered dispatch systems process data on traffic conditions, courier location, specimen stability windows, delivery priority, temperature requirements, and vehicle capacity to generate optimal routing. This produces faster delivery times, fewer missed stability windows, more efficient fleet utilization, and lower operational costs compared to manual dispatch methods.
What should healthcare organizations look for when selecting a medical logistics partner?
Healthcare organizations should evaluate medical logistics partners based on regulatory compliance documentation across HIPAA, OSHA, DOT, and CLIA; temperature management capabilities across all five temperature tiers; technology infrastructure including real-time tracking, digital chain of custody, and AI-powered dispatch; courier training and certification programs; and performance metrics including delivery time reliability, specimen rejection rates, and customer retention. A partner with demonstrated expertise in healthcare logistics provides significantly better outcomes than a general logistics provider.
Related Reading
- Comprehensive Guide to Medical Courier Services
- Why Hospitals Outsource Medical Courier Operations
- STAT Medical Courier Service: Emergency Response Standards
- Medical Specimen Transport: Protocols and Compliance
- HIPAA Compliant Medical Courier: What to Look For
- Get a Quote — carGO Health Medical Courier Services
Related Resources